Rapid microbial diagnostics
A method for rapid detection of microorganisms using chemiluminescence.
Applications
- Microbial screening and identification in food and medical industries
Key Benefits & Differentiators
- High sensitivity:with a limit of detection as low as 100 cfu/mL can be achieved
- Rapid: tests can be performed in 1 hour
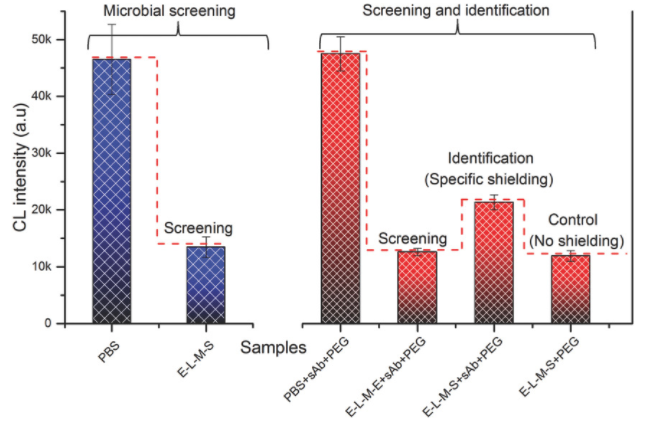
- Broad screening by direct detection of microorganisms; not detecting the byproducts
- Specific testing: using microbial macromolecular shielding
Rapid microbial diagnostics
Rapid detection of microorganisms is vital in the food processing industry to prevent foodborne disease, and in clinical settings for diagnosis and treatment. Traditional methods of detection require the growth of microorganisms in media and detection is only possible when growth reaches a high number of colony-forming units, which can take several days. On the other hand, biomarker-based detection methods are fast but lack specificity and have poor sensitivity. These techniques still require 12-48 hours of enrichment before detection.
Prof. Abdennour Abbas at the University of Minnesota has developed a high-sensitive microbial screening and identification assay that can be completed in 1 hour. Rapid screening of samples is enabled by a chemiluminescence signal that is altered by the presence of microorganisms. The method of action is as follows: the oxidation of gold nanoplates (or nanotriangles, AuNT) particles by tris(2-carboxyehtyl)phosphine (TCEP) generates a long term, stable chemiluminescence signal in the presence of luminol chemical. As a reducing agent, TCEP also targets disulfide bridge (DSB)-containing proteins on the surface of microorganisms. The competitive reaction of TCEP-gold nanoplates and luminol in conjunction with the TCEP reaction with the DSB proteins of microorganisms allowed the researchers to develop methods for screening the presence of microorganisms. Additionally, specific pathogens can be shielded from the chemiluminescence signal by antibodies which allows for identification of target microorganisms. Use of this technology will not only aid in the treatment and prevention of microbial diseases but also reduce costs in the medical and food industry due to increased automation and faster processing times.
Phase of Development
TRL: 3-4Proof of concept. The technique is demonstrated by specific detection of methicillin‐resistant Staphylococcus aureus in environmental and urine samples containing a mixture of microorganisms.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- Abdennour Abbas, PhD, Associate Professor, Bioproducts and Biosystems Engineering
-
expand_more library_books References (1)
- Minh-Phuong Ngoc Bui, John Brockgreitens, Abdennour Abbas , Gold Nanoplate-Enhanced Chemiluminescence and Macromolecular Shielding for Rapid Microbial Diagnostics
-
expand_more cloud_download Supporting documents (1)Product brochureRapid microbial diagnostics.pdf