Multifunctional, flexible sensor array for urethral catheters (20140233)
A new urethral catheter for simultaneous measurement of pressure distribution and neural activity.
Applications
- Urodynamic test for urinary incontinence
- multiple sclerosis, myasthenia, ALS, and other neuromuscular diseases.
- Measurement of pressure in the heart or cardiovascular system
- Anorectal physiologic studies
Key Benefits & Differentiators
- Multiple pressure readings to understand pressure distribution
- Real-time measurement enables testing during provocative maneuvers
- Simultaneous EMG and Pressure measurement using integrated sensor system
- Flexible sensor array allows uninterrupted testing
- Elimination of painful pull-backs needed during study
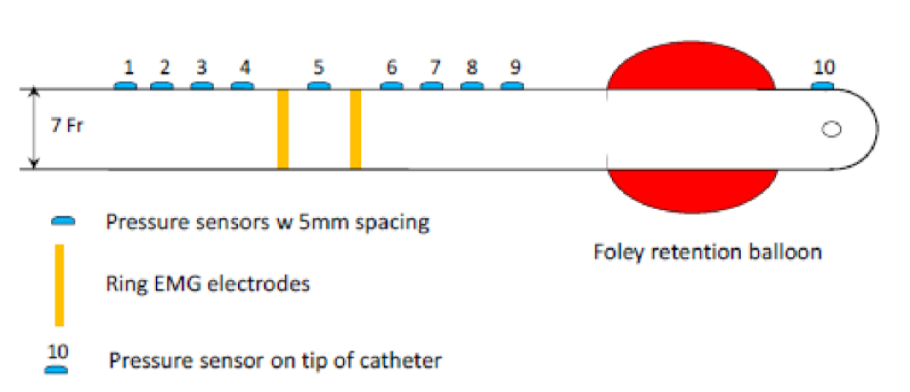
Simultaneous measurement of urethral pressure distribution and EMG
Urodynamic tests are used to diagnose patients who may have urinary incontinence (UI) or other lower urinary tract symptoms. These tests may involve measurement of pressure in and around the urinary bladder (cystometrography), nerve and muscle function (electromyography), and flow rates (uroflowmetry). Currently available catheters for pressure measurements, however, can only measure pressure at a single point in the urethra and cannot be used for real-time measurements. In addition, catheters currently available in the market cannot be used for recording urethral pressure profiles during provocative manoeuvres, such as coughing and val salva.
Researchers at the University of Minnesota have developed a novel sensor system for urethral catheter that is capable of simultaneously measuring pressure distribution in the urethra and neural signals from urethral sphincters. With an array of pressure sensors along its length, and an electromyography (EMG) recording electrode that can be positioned at the urogenital diaphragm, this urethral catheter enables concurrent measurement of distributed urethral occlusive pressures and sphincter EMG. The flexibility and dual-modality of the sensor system enables real-time measurement of urethral pressure distribution during provocative actions such as coughing and val salva maneuvers.
The sensor strip can be significantly bent during urethral insertion into the body. The developed sensor has important clinical applications in urodynamic testing and potentially in other in-vivo biomedical catheter applications. Parasitic capacitance noise expected during measurement procedure is addressed using novel reference measurements. This novel catheter can provide significantly improved urodynamic diagnosis for addressing urinary incontinence, help determine structural and neurological causes of urinary incontinence, provide instantaneous and continuous measurements of functional urethral length, and help in determining appropriate rational procedures for surgical intervention.
Phase of Development
TRL: 4The sensing strip was embedded on a catheter and experimental in-vitro evaluation was conducted using a bench-top pressure chamber.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- Rajesh Rajamani, PhD Professor, Mechanical Engineering
- Gerald Timm, PhD Professor, Department of Urology
-
expand_more library_books References (1)
- Ahmadi, Mahdi, Rajesh Rajamani, Gerald Timm, and A. Serdar Sezen. , Flexible distributed pressure sensing strip for a urethral catheter, Journal of Microelectromechanical Systems
-
expand_more cloud_download Supporting documents (1)Product brochureMultifunctional, flexible sensor array for urethral catheters (20140233).pdf