Minimally invasive diagnostic for osteosarcoma in dogs
A panel of six definitive biomarkers that can be isolated from a canine blood sample for the detection of minimal residual disease and monitoring remission in dogs treated for osteosarcoma.
Applications
- Monitoring remission and relapse of canine cancers
- Diagnosis of Osteosarcoma in dogs
- Routine screening in veterinary practice
Key Benefits & Differentiators
- Improved prognosis for canines with osteosarcoma: Detection of minimal residual disease during treatment to personalize therapy and improve quality of life.
- Minimally invasive and simple: Biomarkers can be detected with only a blood sample. Current methods to effectively monitor osteosarcoma following diagnosis require regular physical exams, PET-CT, and/or bone scans, which are incredibly costly and inconvenient.
- Sensitive and specific: Biomarker signature was developed to differentiate dogs with residual osteosarcoma from healthy animals, as well as from animals with other cancers or illnesses.
- Novel early diagnostic potential: Potential to facilitate earlier diagnosis of osteosarcoma. Current methods that rely on the presence of clinical signs, at which point more than 90% of dogs have micrometastatic disease.
Identifying canine osteosarcoma’s fingerprint
Osteosarcoma is the most common primary bone tumor found in dogs, particularly in large breeds where occurrence can be as high as 20%. Although effective treatment is available if the cancer is caught early (amputation, irradiation, chemotherapy) there are no safe, early diagnostic tests. Furthermore, osteosarcomas are highly aggressive cancer and micrometastasis occurs in up to 90% of dogs, emphasizing the importance of close monitoring following treatment. Unfortunately, the current methods to assess dogs for relapse are multiple radiographs or CT scans every three to six months respectively. There is a clear need for a simple, low risk method to detect canine osteosarcoma for use in diagnostics as well as after diagnosis and/or remission in order to inform treatment decisions in a timely manner.
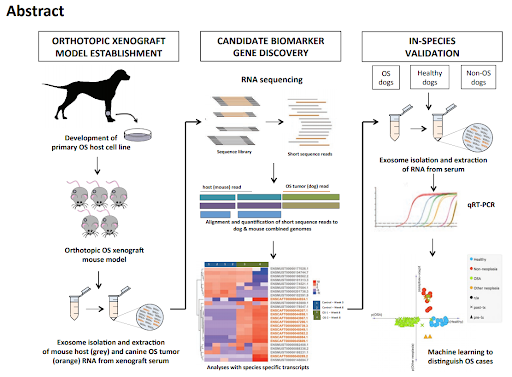
Dr. Jaime Modiano’s lab at the University of Minnesota used a method they previously developed (UMN Case No. 20170056) to identify a differential biomarker signature for osteosarcoma in dogs. The biomarker consists of a panel of exactly six, cell free nucleic-acid biomarkers that can be collected from a simple, minimally invasive blood sample. The biomarkers were identified using samples collected from dogs with osteosarcoma as compared to dogs with no evidence of disease, other tumors or other diseases in order to create a test that was sensitive and specific for osteosarcoma. Validated for the detection of residual osteosarcoma following treatment, this biomarker is currently also being developed and tested for use as an early, minimally invasive diagnostic.
Phase of Development
TRL: 3-4Biomarker panel developed and validated using 62 dogs, including dogs with osteosarcoma, other tumors, other conditions, and dogs with no evidence of disease.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- Jaime Modiano, VMD, PhD Perlman Professor of Oncology and Comparative Medicine, Department of Veterinary Clinical Sciences
-
expand_more library_books References (1)
- Development of an exosomal biomarker signature to detect minimal residual disease in dogs with osteosarcoma using a novel xenograft platform and machine learning (2021), Kelly M. Makieslski et al
-
expand_more cloud_download Supporting documents (1)Product brochureMinimally invasive diagnostic for osteosarcoma in dogs.pdf