First-generation degraders for class I bromodomain-containing proteins
A bromodomain-targeting degrader of CECR2, an epigenetic regulator that drives cancer metastasis.
Applications
- Anti-cancer therapy
- Research tool
Key Benefits & Differentiators
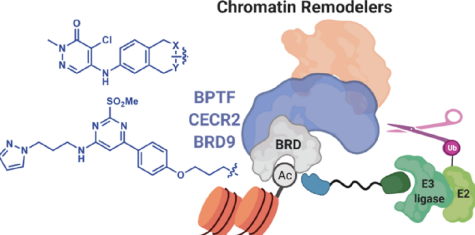
- Targeted protein degradation: The technology enables targeted degradation of specific bromodomains, particularly class I (BPTF and CECR2) and IV (BRD9) bromodomains.
- Scaffold diversity and selectivity: The utilization of two distinct scaffolds derived from pyridazinone and pyrimidine-based heterocycles allows for scaffold diversity in the design of degraders.
Technology Overview
Metastasis is the major cause of cancer-related deaths due to the lack of effective therapies. Emerging evidence suggests that certain epigenetic and transcriptional regulators drive cancer metastasis, either of which could be targeted as a therapeutic strategy. CECR2, a class I bromodomain-containing epigenetic regulator, has recently been identified as a requirement for breast cancer metastasis in mouse models. CECR2 increases the expression of metastasis-associated signaling molecules, and the deletion or inhibition of CECR2 prevents metastasis, suggesting that it is a promising target for anti-cancer therapy. Targeted protein degradation is an emerging technology that can be used for modulating the activity of epigenetic protein targets. However, very few degraders have been reported for class I bromodomain-containing proteins such as CECR2, demonstrating an unmet need.
Researchers at the University of Minnesota have developed first-generation degraders for class I bromodomain-containing proteins, including CECR2, based on two scaffolds derived from pyridazinone and pyrimidine-based heterocycles. Various exit vectors and linkers were evaluated to identify analogues that
demonstrate selectivity for this class of bromodomains. An intracellular assay was used to demonstrate that these heterobifunctional molecules are cell-permeable, form ternary complexes, and can degrade nanoluciferase−bromodomain fusions. As a first example of a CECR2 degrader, the pyrimidine-based analogues degrade endogenous CECR2 in a dose-dependent manner, with almost complete degradation at concentrations between 5 and 50 nM. These degraders could potentially provide a starting point to evaluate the effects of targeted protein degradation in cancer models, and they demonstrate commercial potential as anti-cancer therapeutics.
Phase of Development
TRL: 3-4Researchers have generated in vitro data demonstrating the efficacy of the degrader against CECR2.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- William C.K. Pomerantz, PhD Professor, Department of Chemistry
-
expand_more library_books References (1)
- Huda Zahid, Jeff P. Costello, Yao Li, Jennifer R. Kimbrough, Marisa Actis, Zoran Rankovic, Qin Yan, and William C. K. Pomerantz (2023), Design of Class I/IV Bromodomain-Targeting Degraders for Chromatin Remodeling Complexes, ACS Chem. Biol., 18, 1278−1293
-
expand_more cloud_download Supporting documents (1)Product brochureFirst-generation degraders for class I bromodomain-containing proteins.pdf