Surface markers for identification and isolation of skeletal muscle stem cells
A list of cell surface marker antibodies that can identify and isolate (or purify) human skeletal muscle stem cells.
Applications
- Purification of skeletal muscle stem cells from muscle biopsies for study of novel compounds
- Purification of skeletal muscle stem cells from teratomas.
- Purification from in vitro differentiated iPS cells.
- Regenerative therapy for muscle damage or diseases, i.e. muscular dystrophy.
Key Benefits & Differentiators
- Enhanced purity and enrichment of stem cells: No contamination with other cells, which is observed in traditional technique of fractionating based on differential adherence
- No recombinant selection markers: Native cell surface proteins are used with monoclonal antibodies for effective FACS precluding the use of GFP or other recombinant markers
Technology Overview
Muscular dystrophy (MD) is a group of hereditary diseases characterized by progressive weakness and loss of muscle mass. MD affects about 250,000 people in the United States. Regenerative medicine offers promise in the treatment of MD. However, the use of pluripotent stem cells as a therapeutic intervention is limited by the inability to obtain pure skeletal muscle stem cells suitable for implantation without the use of genetic markers. Additionally, current fractionation methods based on differential cell adherence have significant limitations since they are often contaminated with other cells. The advancement of MD stem cell therapy to the clinic requires novel methods and processes for the identification and purification of muscle stem cells from biopsies or other suitable sources, such as teratomas.
Researchers at the University of Minnesota have identified several surface proteins that are expressed on biopsy-derived or teratoma-derived skeletal muscle stem cells. These surface markers are suitable for both positive and negative selection to ensure yield purity of stem cells and have been used in animal studies. Briefly, teratomas generated in mice were used to produce large numbers of alpha7-Integrin+ and VCAM-1+ myogenic progenitors. These stem cells were FACS purified and transplanted in the diseased muscle where they exhibited high engraftment potential. The newly generated muscle fibers express adult myosin, improve muscle contractile force, and have quiescent PAX7+ stem cells. The PAX7+ stem cells could enable long-term muscle regeneration. These teratoma-derived skeletal muscle stem cells are of similar regenerative potential in comparison to the adult stem cells for skeletal muscle. Together, purified skeletal muscle stem cells can be used to study their therapeutic applications, or to study novel compounds for MD treatment.
Phase of Development
TRL: 3-4Animal studies showing engraftment and muscle regeneration.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- Michael Kyba, Ph.d. Professor, Department of Pediatrics
- Sunny Sun-Kin Chan, Ph.d. Assistant Professor, Department of Pediatrics
Legend for Image
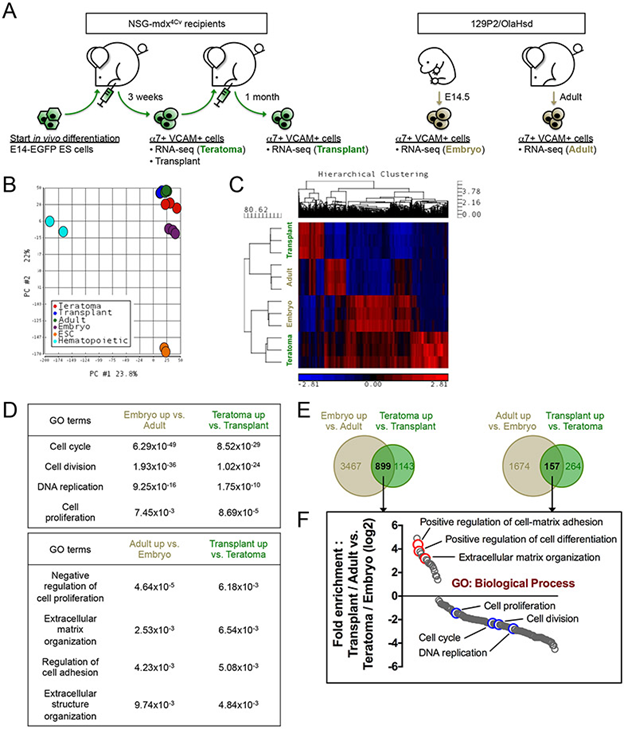
(A) Schematic of samples used for transcriptome analysis. α7+ VCAM+ myogenic cells were isolated from E14-ES cell teratomas (Teratoma), transplanted TA muscles (Transplant), E14.5 embryos (Embryo) and 8-week-old adult hindlimbs (Adult) for RNA-seq (n=3 biological replicates). (B) Principal Component Analysis (PCA). The transcriptomes of the 4 α7+ VCAM+ myogenic populations (Teratoma, Transplant, Embryo and Adult) are very similar to each other, in comparison with those of ES cells and hematopoietic progenitors (from ENCODE (encodeproject.org)). (C) Hierarchical clustering of differentially expressed genes demonstrates a transcriptome similarity between Transplant myogenic cells and Adult satellite cells, and between Teratoma cells and Embryo progenitors. (D) Gene Ontology (GO) Biological Process terms denoting genes enriched in the Embryo-Adult comparison (left column) and the Teratoma-Transplant comparison (right column). p-values of the GO terms are indicated. (E) Venn diagrams showing differential genes commonly upregulated by Teratoma / Embryo progenitors and Transplant / Adult cells. The number of differential genes is indicated. (F) Fold enrichment analysis of genes obtained from (E) based on GO Biological Process. α7, α7-integrin. VCAM, VCAM-1. ES cells / ESC, embryonic stem cells. GO, Gene Ontology.-
swap_vertical_circlelibrary_booksReferences (1)
- Chan, Sunny Sun-Kin, Robert W. Arpke, Antonio Filareto, Ning Xie, Matthew P. Pappas, Jacqueline S. Penaloza, Rita CR Perlingeiro, and Michael Kyba , Skeletal muscle stem cells from PSC-derived teratomas have functional regenerative capacity
-
swap_vertical_circlecloud_downloadSupporting documents (1)Product brochureSurface markers for identification and isolation of skeletal muscle stem cells.pdf