High-throughput screening assay for sickle cell disease drug discovery
A high-throughput screening assay for discovering small molecules that directly inhibit sickle hemoglobin oligomerization.
Applications
- Sickle cell drug discovery
- Sickle cell disease research
Key Benefits & Differentiators
- High-sensitivity: Assay uses high-sensitivity fluorescence lifetime (FLT) FRET
- High-throughput: Assay uses FLT plate reader technology enabling high-throughput screening
Technology Overview
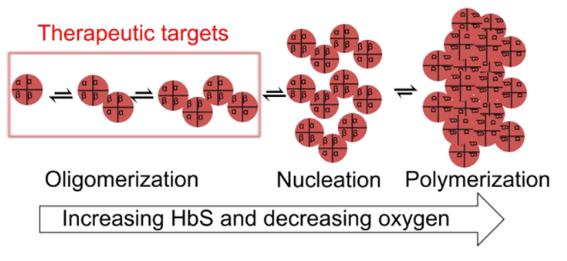
Sickle cell disease (SCD) is a devastating hereditary disorder caused by a defect in the hemoglobin gene that leads to sickle hemoglobin (HbS) polymers that distort the shape of red blood cells. Despite contributing to a shortened lifespan, high infant mortality rates, and significantly reduced quality of life, treatment options for SCD patients are limited to four FDA-approved therapies that do not dramatically improve outcomes. While gene therapy has elicited considerable excitement, the need for bone marrow ablation and the high cost preclude broad application of this approach. Millions of SCD patients worldwide would benefit from new, affordable, and easily accessible therapies. Thus, new small molecule approaches are desperately needed, but the pipeline for identifying and validating new therapies is extremely limited.
After observing that deoxygenated HbS forms temporally stable oligomers at sub-nucleation concentrations, researchers at the University of Minnesota developed a high-throughput screening (HTS) assay for discovering small molecules that directly inhibit HbS oligomerization. This FRET-based assay leverages high-sensitivity fluorescence lifetime measurements that monitor the temporally stable HbS oligomers. In the lab, the assay demonstrated HbS oligomer sensitivity to known polymerization inhibitors and identified novel HbS oligomerization inhibitors that reduced hypoxia-induced impairment of sickle blood flow without increasing hemoglobin affinity. This screening platform has commercial potential for large-scale HTS of small molecules that could be developed into new therapeutics for SCD.
Phase of Development
TRL: 4-5High-throughput screening assay demonstrated in the lab HbS oligomer sensitivity to known polymerization inhibitors and identified novel HbS oligomerization inhibitors.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- David Wood, PhD Associate Professor, Department of Biomedical Engineering
- Jonathan Sachs, PhD Professor, Department of Biomedical Engineering
-
expand_more library_books References (1)
- Nagamani VunnaNagamani Vunnam , Scott Hansen, Dillon C. Williams, MaryJane Olivia Been, Chih Hung Lo, Anil K. Pandey, Carolyn N. Paulson, John A. Rohde, David D. Thomas, Jonathan N. Sachs, and David K. Wood (2022), Fluorescence Lifetime Measurement of Prefibrillar Sickle Hemoglobin Oligomers as a Platform for Drug Discovery in Sickle Cell Disease, Biomacromolecules, 23, 9, 3822-3830
-
expand_more cloud_download Supporting documents (1)Product brochureHigh-throughput screening assay for sickle cell disease drug discovery.pdf