Broadspectrum potentially therapeutic nanobodies against COVID-19
A class of nanobodies (Nanosota-2, -3, and -4) offering broadspectrum therapeutic potential against COVID-19.

Applications
- Antiviral Therapy
- Research Tool
Key Benefits & Differentiators
- High Potency: These nanobodies exhibit strong antiviral activity, effectively neutralizing SARS-CoV-2 and its variants, leading to reduced disease severity.
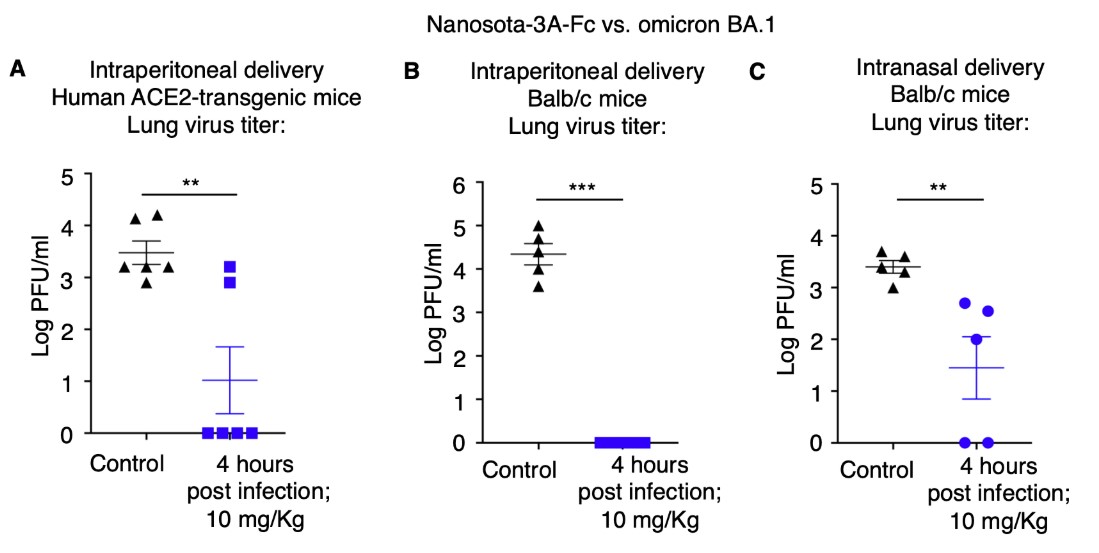
- Broad Spectrum Activity: These nanobodies target various SARS-CoV-2 strains, including major variants like omicron, and uniquely address both SARS-CoV-1 and SARS-CoV-2.
- Ease of Administration: Nanobodies offer convenient delivery options, including inhalation, improving patient compliance and deployment feasibility.
Technology Overview
The COVID-19 pandemic underscored the urgent need for effective antiviral therapeutics capable of combating SARS-CoV-2 and its evolving strains. With new variants emerging, there is a critical demand for innovative treatment options that can address the limitations of existing approaches. Conventional antibodies, while commonly used in therapeutic interventions, encounter significant challenges that hinder their effectiveness in combating COVID-19. These challenges include high production costs, limited potency across variants, and the necessity for injection-only administration, which can impede widespread deployment and accessibility of treatment. There is an increasing imperative for novel solutions that can offer enhanced efficacy, affordability, and ease of administration.
Researchers at the University have developed the Nanosota series—a class of nanobodies offering superior therapeutic potential against COVID-19. Nanobodies, characterized by their small size and exceptional therapeutic properties, have emerged as promising candidates for antiviral therapy. The Nanosota series comprises three anti-SARS-CoV-2 nanobodies—Nanosota-2, -3, and -4—derived from alpacas immunized with the SARS-CoV-2 spike protein. These nanobodies exhibit potent inhibition of viral infection, with Nanosota-2 demonstrating particularly impressive efficacy, inhibiting prototypic SARS-CoV-2 infection in vitro at low IC50 concentrations and in mice at low dosages or late administration times. Additionally, Nanosota-3 displays potent activity against the omicron variant, while Nanosota-4 uniquely targets both SARS-CoV-1 and SARS-CoV-2. The Nanosota series represents a significant advancement in COVID-19 therapeutics, offering super potency and a wide spectrum of activity against SARS-CoV-2 and its major variants, with the potential for cost-effective production, adaptation to new viral variants through phage display, and deployment via inhalation delivery.
Phase of Development
TRL: 3-4Researchers have obtained preclinical data using these nanobodies.
Desired Partnerships
This technology is now available for:- License
- Sponsored research
- Co-development
Please contact our office to share your business’ needs and learn more.
Researchers
- Fang Li , PhD Professor, Edmund Wallace Tulloch and Anna Marie Tulloch Endowed Chair, Director of Center for Coronavirus Research
- Bin Lui, PhD Associate Professor, The Hormel Institute
-
expand_more library_books References (1)
- Gang Ye, Ruangang Pan, Fan Bu, Jian Zheng, Alise Mendoza, Wei Wen, Lanying Du, Benjamin Spiller, Brian E. Wadzinski, Bin Liu, Stanley Perlman, Fang Li , Discovery of Nanosota-2, -3, and -4 as super potent and broadspectrum therapeutic nanobody candidates against COVID-19, Journal of Virology
-
expand_more cloud_download Supporting documents (1)Product brochureBroadspectrum potentially therapeutic nanobodies against COVID-19.pdf